This pilot in vitro study explored how decompression-induced microbubble nucleation and acoustic softening contribute to flow instability and potential SFC in human blood. The experimental setup simulated pressure conditions relevant to DCS, especially during high-altitude exposure and spaceflight, offering new insights into the compressible behavior of blood under stress. A visual sequence captured during the experiment demonstrated progressive microbubble nucleation and distinct phase transitions in venous blood as it was subjected to vacuum-induced decompression ranging from 350 to 650 mmHg at 40 °C (see Figs. 1, 2). Prior to the vapor pressure test, the blood sample appeared stable, while post-test observations revealed significant gas evolution and structural changes. A video of the full vapor pressure test is available at: https://www.youtube.com/watch?v=pHStIayAYas.
Fig. 1: Microbubbles in venous blood of healthy humans under reduced pressure.
a–e Magnified views of microbubble nucleation and associated textural changes in venous blood samples collected from different healthy adult individuals, exposed to vacuum-induced decompression (350–650 mmHg at 40 °C). All images depict visually detectable microbubble formation, with notable inter-subject variation in bubble distribution, density, and structural transitions. These variations reflect individual-specific thermophysical responses of blood under decompressive stress, offering insight into the morphological pathways leading to potential cardiovascular risks in extreme environments such as spaceflight.
Fig. 2: Subject-specific differences in microbubble formation under vacuum-induced decompression.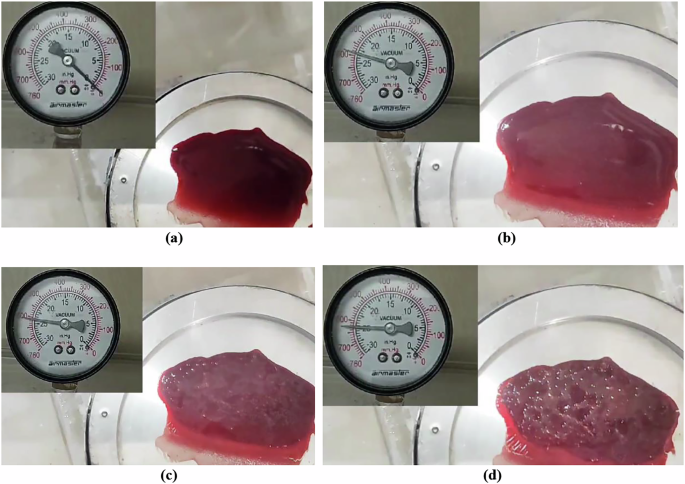
a–d Progressive stages of visually detectable bubble nucleation and phase transitions in venous blood during vacuum-induced decompression at 40 °C, showing: a fresh blood sample before devacuumization, b initial observable bubbling at 590 mmHg, c increased bubble intensity at 610 mmHg, and d advanced aggregation and structural transitions at 625 mmHg.
Bubble nucleation and acoustic softening–induced Sanal flow choking
Gradual decompression of fresh venous blood samples at physiological temperatures (37–40 °C) led to reproducible microbubble formation, which first became visible between 590 and 625 mmHg [Fig. 2a–d]. As pressure continued to decrease, nucleation intensified, suggesting the presence of a vapor pressure threshold that initiates bubble formation. Repeated trials confirmed consistent nucleation behavior at pressures below ~170 mmHg. Surface texture was found to play a key role in bubble formation: rough-threaded surfaces promoted earlier and denser bubble nucleation compared to smooth ones, supporting classical crevice-nucleation theory26. Preliminary observations also suggested subtle gender-based differences in nucleation dynamics, though further investigation is required to validate this finding38.
High-speed imaging revealed a series of progressive thermal and phase changes in blood during decompression. Initial plasma outgassing was followed by increasingly vigorous bubbling, indicating evaporation. At more extreme pressure drops, visual signs of protein denaturation and polymerization emerged, including crust formation at the sample edges. Ultimately, near-complete moisture loss led to a brittle, coagulated state, mimicking tissue behavior under severe decompression conditions. These results (Figs. 1–3) illustrate a clear sequence of decompression-induced phase transitions, with potential relevance to the structural and functional integrity of blood under extreme environmental stress.
Fig. 3: Blood evaporation rate and bubble formation intensity in a 26-year-old healthy male (O+ blood type) during vacuum-induced decompression at 40 °C.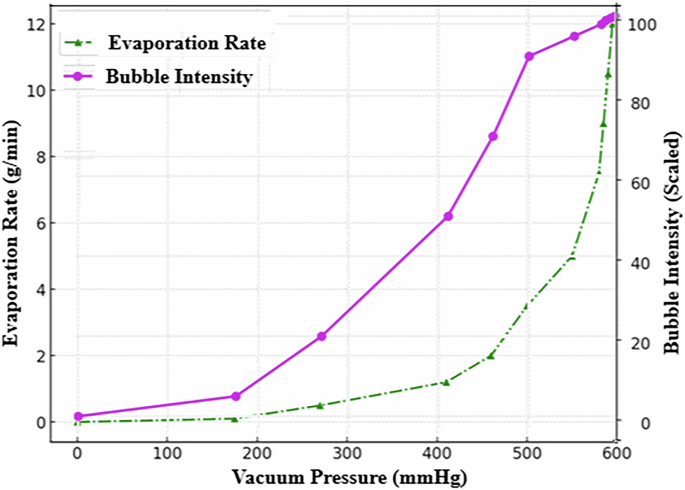
The dual-axis plot shows the relationship between vacuum pressure (mmHg) and two key responses: evaporation rate (g/min, green dashed line with triangles) and scaled bubble formation intensity (magenta solid line with circles). Both parameters exhibit nonlinear increases as ambient pressure decreases, with a pronounced rise in activity between 500 and 600 mmHg. This critical pressure range marks the onset of phase transitions and microbubble nucleation, offering insights into decompression-induced cardiovascular risks.
To validate chamber performance and benchmark fluid behavior, parallel decompression experiments were conducted using water and hydrocarbon fuels. Water drawn from the Narmada River exhibited expected boiling behavior between 545 and 560 mmHg, confirming the reliability of the vacuum decompression system. Narmada water was selected due to its moderate mineral content and regional availability. While this freshwater sample served to validate the experimental setup, future comparisons using seawater—given its higher salinity and dissolved gas content—may offer additional insights into pressure-dependent nucleation behavior.
Petrol, diesel, and kerosene displayed nucleation patterns consistent with their thermophysical properties35,36,37,39. Petrol, with the highest vapor pressure, nucleated earliest, while kerosene exhibited the greatest resistance to bubble formation. These control tests affirmed that fluids with lower heat capacity and higher vapor pressure are more susceptible to early phase transitions under decompression. These findings supported the experimental framework used to investigate blood behavior and provided a reference for interpreting nucleation onset and thermal responses.
Microgravity and space radiation are among the most critical environmental stressors encountered during space missions, with well-documented effects on cellular function, fluid dynamics, and vascular physiology. However, opportunities to study these effects directly in space remain limited due to high mission costs and constraints on experimental repeatability. To address this, ground-based microgravity simulators have been widely used to replicate altered gravity conditions and examine biological responses at the cellular and systemic levels40. Complementing these approaches, the present study introduces a vacuum-based test protocol to simulate another key aspect of spaceflight: pressure variation and decompression stress, which can significantly influence blood behavior. By demonstrating how microbubble nucleation, acoustic softening, and SFC6 occur under controlled vacuum conditions, our findings offer a novel and physiologically relevant method for assessing cardiovascular risks associated with spaceflight-induced decompression events—providing an important bridge between microgravity simulation and in situ physiological modeling.
The study confirmed that the phenomenon of SFC6,7 in cardiovascular system is closely associated with a reduction in sound velocity in bubbly blood flows. As gas bubbles form and disperse, they decrease the speed of sound, leading to compressibility effects and potential shock wave generation. Oscillating bubbles further dissipate energy, reducing sound speed, particularly at resonance frequencies. When gas concentration increases (see Fig. 3), the sound velocity in blood may approach the velocity of blood flow itself, leading to shock wave formation and cardiovascular disruptions such as embolism and hemorrhage, especially in extreme environments like space travel or rapid decompression scenarios.
The observations from this study have significant implications for understanding the risks associated with DCS and cardiovascular health in extreme environments. The formation and behavior of microbubbles under reduced pressure conditions can lead to flow choking, shock wave generation and potential cardiovascular disruptions. Understanding these mechanisms is crucial for developing effective decompression protocols and protective measures for individuals exposed to rapid pressure changes, such as divers and astronauts.
The study corroborates SFC as a novel concept in cardiovascular physiology6, at different physical situations, demonstrating that when the internal-to-external pressure ratio of tiny bubbles exceeds a critical threshold, their rupture can trigger streamtube expansion, leading to significant shock wave formation. This suggests that blood’s compressibility under extreme conditions plays a pivotal role in the development of DCS and other cardiovascular disorders. Furthermore, the study confirms that fluids with higher vapor pressures and lower heat capacities are more susceptible to early nucleation under decompression. This finding underscores the importance of understanding the thermophysical properties of biofluids in assessing the risks associated with rapid decompression scenarios.
This pilot investigation confirms, on first-law grounds, that every moving fluid—including blood—is inherently compressible, because its enthalpy necessarily exceeds its internal energy; therefore, haemodynamic studies that aim to capture SFC must incorporate compressible-flow effects, especially under decompression. Here we demonstrate that a rapid pressure drop initiates a coupled cascade of microbubble nucleation, acoustic softening, and SFC in whole blood. Reproducible vacuum-gauge thresholds marked a sequence from plasma outgassing through evaporation and protein polymerization to final coagulation, and high-speed imaging revealed transient shock-like fronts whenever local Mach numbers approached unity. Rough-textured coupons triggered earlier, denser bubbling than polished controls, underscoring surface micro-geometry as a key design variable for future cardiovascular devices.
Although absolute sound speed was inferred rather than measured, the void-fraction–dependent softening predicted by Wood’s equation aligns with the observed flow perturbations and offers a mechanistic bridge between nitrogen supersaturation and the abrupt vascular events reported in DCS. The parallel tests in water, petrol, diesel, and kerosene validate our pressure-ramp protocol and situate blood within a wider thermophysical landscape. These findings establish a tractable bench model—bio-shock-wave mechanics—for exploring how pressure transients in spaceflight, high-altitude aviation, or deep-sea ascent can compromise endothelial integrity, myocardial perfusion, and overall haemodynamic stability. They also point to practical countermeasures: smoother implant surfaces, revised prebreathe schedules, and real-time monitoring of acoustic properties during EVA.
Future work should (i) directly measure sound speed and pressure fields in pulsatile, endothelialised flow systems, (ii) map blood heat capacity and vapor pressure across diverse clinical cohorts to refine flow choking thresholds, and (iii) integrate high-fidelity bubble metrics into AI-driven wearables for early risk prediction. Addressing these gaps will convert the present proof-of-concept into an operational toolset for safeguarding cardiovascular health in extreme environments.
This pilot investigation has several limitations that future work should address. First, acoustic softening was inferred rather than measured; direct, high-fidelity quantification of sound speed, void fraction, and bubble kinematics is needed. Techniques such as digital inline holography and three-dimensional particle-tracking velocimetry could capture bubble size, shape, density, and trajectories, while high-speed confocal or phase-contrast microscopy would reveal membrane thickness, rupture modes, and wall adhesion. Complementary laser-induced fluorescence and Raman spectroscopy could identify the evolving gas composition inside and around bubbles.
Real-time mapping of void fraction and microbubble intensity is another critical gap. X-ray phase-contrast imaging, optical coherence tomography, and ultrasound backscatter analysis can supply high-resolution distributions of microbubbles in flowing blood analogs or vascular constructs, generating richer datasets for AI-driven decompression-risk models.
Quantifying the exact pressure ratio that triggers SFC in anatomical geometries will require pressure-sensitive microchannels fitted with micro-PIV and embedded MEMS pressure sensors. Such instrumentation can pinpoint the threshold at which choking and shock-like jumps begin.
Bubble nucleation is also highly sensitive to surface microtexture and radiation-induced changes in material chemistry. Atomic-force microscopy and three-dimensional profilometry should be used to characterize roughness, whereas controlled irradiation studies can reveal how space-relevant radiation alters surfaces, blood chemistry, membrane stability, and thermophysical properties that influence nucleation and choking.
An integrated, AI-ready data pipeline is essential. Future experiments must systematically capture bubble morphology, growth kinetics, spatial void-fraction fields, gas composition, surface correlations, critical pressure ratios, Mach-number evolution, and the roles of temperature, viscosity, heat capacity, and geometry under variable pressure, radiation, and microgravity conditions.
Ultimately, real-time, in-vivo monitoring will depend on miniaturized optical, acoustic, or magneto-elastic sensors capable of detecting microbubble onset, acoustic-property shifts, local pressure gradients, and velocity changes indicative of choking. Coupling these data streams to adaptive AI models that reference individual physiological baselines could give astronauts early warning of decompression stress, enabling preventive action before symptomatic DCS or cardiovascular compromise develops. Addressing these limitations with advanced diagnostics will advance a real-time cardiovascular-risk surveillance framework for deep-space missions, uniting experimental fluid physics, bioengineering, and artificial intelligence to safeguard crew health.
